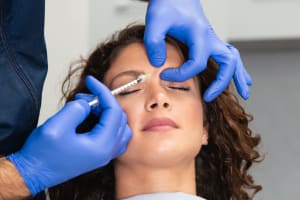
Concerns Treated

Fine Lines & Wrinkles
Smooth as a baby’s bottom. Not saying we necessarily want to correlate that to our faces, but the idea is the same. Fine lines, fine wrinkles, and crepey skin can disrupt the surface of our skin — let’s fix that.
Medshop
Not ready for a procedure? Explore powerful at-home alternatives.
Concerns Treated

Fine Lines & Wrinkles
Smooth as a baby’s bottom. Not saying we necessarily want to correlate that to our faces, but the idea is the same. Fine lines, fine wrinkles, and crepey skin can disrupt the surface of our skin — let’s fix that.
Discover science-backed solutions from AEDIT Medshop
We've taken out the guesswork so you can shop with confidence, knowing you're choosing what's right for you.

To learn more about our content creation practices, visit our Editorial Process page.